What’s in this audio? (Click to expand)
- Introduction: Why injection site choice dominates every Mounjaro forum and pharmacy counter conversation.
- The Clinical Trial Foundation: NCT04050670’s 54-participant crossover study comparing abdomen, thigh, and upper arm with 35-day washout periods.
- The Paradox Explained: How identical total drug absorption (AUC) produces wildly different side effects—44% less nausea with thigh injections.
- Appetite Suppression Trade-offs: Why abdomen shows 4x more reported appetite suppression but adherence matters more than acute effects.
- Practical Site Selection: Two evidence-based strategies—comfort priority (thigh) versus initial effect priority (abdomen with risks).
- Injection Technique Essentials: Approved zones, why inner thigh must be avoided, and the 45° angle pinch technique for lean individuals.
- Rotation Protocol: How failure to rotate creates 8.85x higher lipohypertrophy risk causing erratic absorption and sabotaged therapy.
- Strategic Recommendations: Study limitations, personal experimentation logs, and dynamic site selection as a conscious symptom management tool.
If you’re using Mounjaro, you may wonder if the injection site matters. With conflicting advice online, mostly anecdotal, it’s hard to know what truly works.
We wanted to know what the clinical data actually show. So we dug into the trial results.
We found that while absorption is similar regardless of injection site, the reported adverse-event rates varied by site. One location showed almost half the rate of nausea as another.
We’ve supported hundreds of clients through their Mounjaro journey at our Rotherham clinic, and questions about injection sites come up in almost every consultation. This guide combines the findings of research with our practical observations.
If you’re using Zepbound (the US brand name for tirzepatide), everything here also applies to you. Same medication, same injection guidance. [[1], [2], [6], [7]]
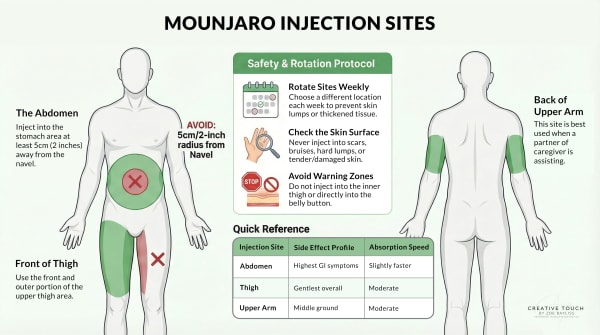
The three approved injection sites
Mounjaro can be injected into three areas: the abdomen, the front of the thigh, or the back of the upper arm. [[1], [6]] Each contains enough subcutaneous fat tissue for proper absorption.
Abdomen (stomach area): Most people find this the easiest site to access. The abdomen typically offers the largest injection area with consistent subcutaneous fat. You’ll need to stay at least 5cm (about 2 inches) away from your belly button, [3] but otherwise have plenty of space to work with.
Front of thigh: The second most popular choice for self-injection. You’ll use the front, outer portion of your upper thigh. It’s easy to see what you’re doing, and most people have adequate fat tissue here.
Back of upper arm: This site usually requires someone else to help with the injection, since it’s difficult to reach on your own. The back of the upper arm typically has more subcutaneous fat than the front, making it suitable for injection when you have assistance.
Most of our clients self-inject into either the abdomen or the thigh. The arm tends to work better when a partner or family member is helping.
What the clinical trial data actually shows
Most advice about Mounjaro injection sites is based on general principles or personal experience. But there is actual clinical trial data comparing the three sites directly.
Clinical trial NCT04050670 examined the relative bioavailability (AUC and Cmax) and tolerability of a single 5 mg tirzepatide dose injected subcutaneously into abdomen, thigh, and upper arm. The study used a randomized, 3-period crossover design in ~54 participants, with a single 5 mg dose given in each period at different sites (abdomen, thigh, upper arm) separated by washouts. [4]
The researchers measured two things: how much medication reached the bloodstream (pharmacokinetics), and what side effects occurred at each site. They compared drug exposure across injection sites using AUC and Cmax. Overall exposure was broadly comparable across sites, although peak levels (Cmax) were modestly lower with thigh and upper-arm injections than with abdomen (details in the Appendix).
But the side effect data told a different story.
Gastrointestinal side effects by injection site
The most striking differences appeared in gastrointestinal symptoms, which are the most common Mounjaro side effects: [4]
Nausea affected:
- 37.04% of participants when injecting into the abdomen
- 22.22% when injecting into the upper arm
- 20.75% when injecting into the thigh
That’s a 44% relative reduction in nausea when using the thigh compared to the abdomen.
Vomiting showed an even more dramatic pattern:
- 16.67% with abdominal injections
- 18.52% with upper arm injections
- 7.55% with thigh injections
Thigh injections more than halve the vomiting rate compared to the abdomen.
Indigestion (dyspepsia) followed a similar trend:
- 18.52% with abdomen
- 20.37% with upper arm
- 11.32% with thigh
Other gastrointestinal side effects - constipation, diarrhoea, and flatulence - were reported less often with thigh injections as well. [4]
Appetite suppression differences
Here’s an important nuance. Decreased appetite is one of the key mechanisms behind Mounjaro’s weight loss effects. In this trial, the symptom label ‘decreased appetite’ was reported at different rates depending on where the injection was given: [4]
- Abdomen: 24.07% reported decreased appetite
- Upper arm: 12.96% reported decreased appetite
- Thigh: 5.66% reported decreased appetite
In the study’s side-effect reporting, ‘decreased appetite’ was recorded approximately four times more often with abdomen injections than with thigh injections. If reducing appetite is your primary goal, this seems significant, but it’s still a side-effect report and not a direct measure of weight-loss results or medication ‘strength.’
In the same dataset, abdomen injections also had higher reported nausea than the other sites, so some people may prefer a site that feels gentler even if it reports ‘decreased appetite’ less often.
Put simply: in this study, ‘decreased appetite’ is being counted as a reported symptom (an adverse-event term), not used as proof that one injection site works better for weight loss.
Other side effects
Headache rates also varied:
- 22.22% with abdomen
- 14.81% with upper arm
- 11.32% with thigh
In this dataset, the thigh had lower reported rates across the majority of adverse events measured in the trial. [4]
| Side Effect (Symptom) | Abdomen (%) | Upper Arm (%) | Thigh (%) |
|---|---|---|---|
| Nausea | 37.0 | 22.2 | 20.8 |
| Vomiting | 16.7 | 18.5 | 7.6 |
| Indigestion (Dyspepsia) | 18.5 | 20.4 | 11.3 |
| Burping (Eructation) | 5.6 | 7.4 | 1.9 |
| Headache | 22.2 | 14.8 | 11.3 |
| Decreased Appetite | 24.1 | 13.0 | 5.7 |
The pharmacological puzzle
Here’s the part that’s genuinely puzzling. The trial found broadly comparable systemic exposure across abdomen, thigh and upper arm, meaning the body absorbs a similar overall amount of tirzepatide regardless of site. [4]
So why would side effects differ if overall exposure is similar? [4]
We don’t have a definitive answer. The researchers didn’t explain this discrepancy, and no follow-up studies have investigated it. But there are a few possible explanations:
Absorption rate vs total absorption: One clue comes from the pharmacokinetics. While overall exposure (AUC) was broadly comparable across sites, peak concentrations (Cmax) were modestly lower with thigh and upper‑arm injections compared with abdomen. That pattern is consistent with a difference in absorption rate rather than the total amount absorbed, potentially influenced by site factors such as local blood flow/perfusion and tissue uptake dynamics. If side effects are partly “peak-driven”, a higher abdominal peak could plausibly trigger more early GI symptoms even when total exposure is similar.
Local Gut Receptor Activation (“The Leakage Theory”): While Mounjaro mimics the GLP-1 hormone naturally produced by your body’s L-cells, it is administered differently. Natural GLP-1 is released directly in the gut. One untested hypothesis is that injecting into abdominal fat - which sits in close proximity to the enteric nervous system (the “second brain” in your gut) - may allow the medication to “leak” or transport locally to gut receptors before it even enters your general circulation. This could trigger nausea more aggressively than an injection in the thigh, which is far removed from the digestive tract.
Local tissue effects: The injection site itself may influence how your body responds locally before the medication enters systemic circulation.
Statistical variation: With a small sample, some differences could still be down to chance.
Study design factors: This was a single-dose study at 5mg. Effects might differ with repeated weekly dosing at higher maintenance doses.
What we can say is this: the pattern observed in our clinic broadly matches what the trial data suggests. Clients who switch to thigh injections often report fewer stomach issues. That’s not proof, but it’s consistent.
Does the injection site affect weight loss?
This is probably the question you really want answered. And we need to be honest: the trial didn’t directly measure weight loss outcomes. [4] It measured pharmacokinetics and side effects, not how much weight people lost.
The higher appetite suppression from abdominal injections could theoretically support greater weight loss. But that’s speculation, not evidence. And even if true, the effect would likely be modest compared to other factors.
Here’s what actually determines long-term success with Mounjaro for weight loss: the lifestyle and dietary changes you make while the medication is helping you eat less. Your injection site choice will be insignificant compared to whether you’re building sustainable habits.
The TOWARDS study, UK primary care data, and other research into life after Mounjaro consistently show that medication adherence and lifestyle modification drive outcomes. Not where you put the needle.
Proper injection technique
Getting the technique right matters more than which site you choose. And based on the search queries we see, there’s quite a bit of confusion about the specifics.
Where exactly on the thigh?
Use the front, outer portion of your upper thigh only. The upper third, on the anterior-lateral aspect (front and slightly to the outside). [5]
Avoid the inner thigh. This isn’t just preference. Clinical guidance for subcutaneous injections explicitly states not to use the inner thigh. [5] Three reasons:
- Higher concentration of nerves, making injections more painful.
- Proximity to the femoral artery and vein increases the bleeding risk.
- Variable subcutaneous fat depth raises the chance of accidentally injecting into muscle.
Stick to the front and outer thigh. You’ll have plenty of suitable areas to work with.
How far from your belly button?
At least 5cm, which is about 2 inches. Both measurements appear in different guidance documents, and they’re effectively the same distance.
The area immediately around your navel contains tougher connective tissue that can make injection uncomfortable and potentially affect absorption. Give it a wide berth.
The rest of your abdomen, from about 2.5cm above your pubic bone to just below your lowest rib, and out to the sides, is suitable for injection.
Should you pinch the skin?
This depends on your body composition, and the official prescribing information doesn’t give a universal answer. It states: “Your healthcare provider should tell you if you should pinch the skin before injecting.”
General guidance for subcutaneous injections suggests:
- If you have adequate subcutaneous fat: You may not need to pinch at all with shorter needles.
- If you’re lean: A skin pinch at a 45-degree angle helps ensure the medication goes into fat tissue rather than muscle.
- For arm or thigh injections: These sites typically have thinner subcutaneous layers than the abdomen, so a pinch may be warranted even if you wouldn’t need one for stomach injections.
The Mounjaro pen has a pre-set needle, so you can’t select the length. If you’re uncertain about technique, ask during your next consultation. We always demonstrate proper injection technique with our clients.
What happens if you inject into a muscle?
Mounjaro is designed for subcutaneous injection, meaning into the fat layer beneath your skin. If the needle goes deeper into muscle tissue, several things can happen:
Faster, more variable absorption: Muscle has a richer blood supply than fat tissue, which can speed up how quickly the medication enters your bloodstream. This might intensify side effects.
Increased pain and bruising: Muscle tissue has more nerve endings than subcutaneous fat. You’re more likely to experience discomfort during the injection and bruising afterwards.
Potentially erratic drug levels: The consistent, slow absorption from subcutaneous fat is part of how these medications are designed to work. Intramuscular injection bypasses that.
Prevention is straightforward: use the appropriate technique for your body composition. Lean individuals should use a skin pinch. Avoid pressing too hard. And if you’re concerned, the abdomen typically offers the lowest risk of accidental intramuscular injection because most people have adequate fat there.
Why site rotation matters
The prescribing information for both Mounjaro and Zepbound is clear: “Rotate injection sites with each dose.”
A 2024 meta-analysis examined what happens when people don’t rotate properly. The finding was striking: incorrect injection site rotation was the strongest contributor to lipohypertrophy, with an odds ratio of 8.85. That means people who didn’t rotate were nearly nine times more likely to develop these fatty lumps at injection sites.
Lipohypertrophy might sound like a minor cosmetic issue. It isn’t. These lumps cause erratic, unpredictable drug absorption. For insulin users, this significantly increases the risk of blood sugar swings. For GLP-1 medications like Mounjaro, inconsistent absorption could affect both efficacy and side effects.
How to rotate effectively:
Move at least 1-2cm away from your previous injection site. One practical approach is to divide your injection area into quadrants or zones, use one zone per week, and move systematically (say, clockwise around your abdomen).
Keep a simple record. We recommend clients note their injection site and any side effects for at least the first few months. It helps identify patterns and ensures you’re not unconsciously returning to the same spot.
Our practical recommendations
Here’s what we suggest based on the evidence combined with what we’ve observed supporting clients through their Mounjaro treatment.
If you’re struggling with nausea
Consider switching to thigh injections. The trial data showed roughly half the nausea rate compared to abdominal injections, [4] and this matches what we see in practice.
Give it at least two to three weeks to assess whether you notice a difference. Your body needs time to establish a pattern with the new site.
If appetite suppression is your priority
Abdominal injections showed the highest rates of decreased appetite in the trial data. [4] If you’re finding that Mounjaro isn’t curbing your appetite as much as you’d hoped, and you’re currently using thigh or arm, trying abdominal injections might help.
But go in with realistic expectations. You’re also accepting a higher likelihood of GI side effects. It’s a trade-off.
When stepping up to a higher dose
This is where our clinical experience adds something that the research doesn’t capture. When you’re moving to a higher dose, whether from 2.5mg to 5mg, or 5mg to 7.5mg, or any other increase, consider using thigh injections for at least the first week.
The logic: your body is adjusting to more medication. Using a site associated with fewer side effects gives you a gentler transition. You can always switch back to your preferred site once you know how your body handles the higher dose.
We’ve seen this approach work well for many clients. It’s not in any official guidance, but it makes practical sense, and consistent administration matters more than always using the same site.
Finding what works for you
Individual responses vary considerably. The trial data show population-level trends, but you’re not a population average. You’re you.
The best approach is systematic experimentation. Track how you feel, then try a different site and compare. Keep notes. Look for patterns.
What works for someone else might not work for you, and vice versa.
Limitations of the evidence
We’ve based this guide heavily on NCT04050670 because it’s the only clinical trial directly comparing tirzepatide injection sites. [4] But you should know its limitations:
Small sample size: Only 54 participants. That’s enough to detect large differences but might miss subtler patterns.
Single dose only: The trial used 5mg doses. Whether the same patterns hold at maintenance doses of 10mg or 15mg hasn’t been studied.
‘Overtly healthy’ volunteers: Participants had BMIs from 18.5 to 45, but they weren’t specifically people with diabetes or obesity seeking weight loss. The typical Mounjaro user might respond differently.
No replication: No subsequent studies have confirmed or contradicted these findings. In science, a single study is never the final word.
No weight outcomes: The trial measured side effects and pharmacokinetics. It didn’t track whether the injection site affected weight loss over time.
Longer-term clinical outcomes by injection site have not been established by replicated, longer-duration studies; this study was single-dose and focused on PK/tolerability. [4]
This is the best evidence we have. It aligns with our observations in practice. But it’s not definitive, and future research may change our understanding.
Best practices summary
- All three sites deliver equivalent medication absorption. Your bloodstream receives the same amount of tirzepatide regardless of where you inject.
- Trial data suggest thigh injections may produce fewer GI side effects. Nausea and vomiting rates were roughly halved compared to abdominal injections.
- Abdominal injections showed higher appetite suppression but more side effects. There’s a trade-off to consider based on your priorities.
- Avoid the inner thigh. Use the front, outer portion only.
- Stay at least 5cm (2 inches) from your belly button. The tissue there isn’t ideal for injection.
- Rotate injection sites with each dose. This prevents lipohypertrophy and maintains consistent absorption.
- Consider thigh injections when stepping up to a higher dose. It may ease the transition.
- Track your experience. A simple log of injection sites and side effects helps you identify what works for your body.
Conclusion
Your choice of injection site can affect your Mounjaro experience, even though the medication is absorbed similarly regardless of where you inject. The evidence suggests thigh injections offer a gentler side effect profile, while abdominal injections may provide stronger appetite suppression at the cost of more GI discomfort.
There’s no universally “best” site. The right choice depends on your priorities, your body’s response, and what you can sustain comfortably over months or years of treatment.
If you’re starting Mounjaro or struggling with side effects, experimenting with different injection sites is worth trying. Track what happens. Be systematic. And remember that the injection site is just one variable among many that determine your success.
If you’d like support with your weight loss journey, explore our Mounjaro programme or take our food addiction assessment to understand your relationship with food.
Last updated: Dec 2025 Medical review: This article discusses general information about Mounjaro injection sites. It is not a substitute for personalised medical advice. Always follow the guidance provided by your prescribing healthcare professional.
Frequently Asked Questions
- Is there really a “best” injection site for Mounjaro?
Not definitively, but the evidence suggests thigh injections may offer advantages for many people.
What the clinical trial showed:
- Thigh injections - Lowest rates of nausea, vomiting, and other GI side effects across all measures
- Abdominal injections - Highest appetite suppression but also highest side effect rates
- Upper arm - Falls somewhere in the middle for most outcomes
Your “best” site depends on what you’re prioritising. If minimising side effects matters most, thigh may work better. If you want maximum appetite suppression and can tolerate more GI symptoms, abdomen might suit you.
The honest answer is that individual responses vary. What works brilliantly for one person might not suit another.
- Can I inject Mounjaro into my inner thigh?
No - the inner thigh should be avoided. Use the front, outer portion of your upper thigh only.
Why this matters:
- Higher nerve density - The inner thigh has more nerves, making injections more painful
- Blood vessel proximity - The femoral artery and vein run through this area, increasing bleeding risk
- Variable fat depth - Subcutaneous tissue is less consistent here, raising the chance of injecting into muscle
Clinical guidance from major medical centres is explicit: “Do NOT use the inner thigh.” Stick to the front and slightly outer aspect of your upper thigh, roughly the upper third of the leg.
- Should I pinch my skin when injecting Mounjaro?
It depends on your body composition - there’s no universal answer.
General guidance:
- If you have adequate subcutaneous fat - You may not need to pinch at all
- If you’re lean - A skin pinch at a 45-degree angle helps ensure the medication goes into fat rather than muscle
- For thigh or arm injections - These sites typically have thinner fat layers, so pinching is often helpful even if you wouldn’t need it for stomach injections
The Mounjaro pen has a pre-set needle length, so you can’t adjust this. If you’re uncertain about technique, ask your prescriber to demonstrate during your next appointment - it’s a common question and takes just a moment to show.
- What happens if I accidentally inject into muscle instead of fat?
An occasional intramuscular injection isn’t dangerous, but it’s not ideal and you may notice some differences.
What might happen:
- Faster absorption - Muscle has richer blood supply, potentially causing higher initial drug levels
- More discomfort - The injection itself may hurt more, and you might experience more bruising
- Possibly stronger side effects - The altered absorption pattern could intensify initial symptoms
If you suspect you’ve injected into muscle, there’s no need to panic or seek emergency care. Simply note it, and adjust your technique next time. Using a skin pinch for lean areas and choosing the abdomen (which typically has the most subcutaneous fat) can help prevent this.
If you’re consistently finding injections painful or noticing unusual bruising, mention this at your next consultation.
- If the medication absorbs the same everywhere, why would side effects differ by injection site?
This is genuinely puzzling, and researchers haven’t fully explained it yet.
What we know:
- The same total amount reaches your bloodstream - Clinical trial data confirmed equivalent overall absorption (AUC and Cmax) across all three sites
- Yet reported side effect rates differed substantially - Thigh showed roughly half the nausea rate of abdomen despite identical systemic exposure
Possible explanations include differences in absorption rate (even if total absorption is equal), local tissue effects at the injection site, or simply statistical variation in a relatively small study.
We don’t have a definitive answer. What we can say is that the pattern we observe in our clinic broadly matches what the trial data suggests - clients who switch to thigh injections often report fewer stomach issues.
- Should I switch to thigh injections when increasing my dose?
This is something we often suggest to clients, though it’s not in any official guidance.
The reasoning:
- Dose increases often bring temporary side effects - Your body needs time to adjust to higher medication levels
- Thigh injections showed lower GI side effect rates - Using this site during the adjustment period may ease the transition
- You can always switch back - Once you know how your body handles the new dose, return to your preferred site if you wish
Try thigh injections for at least the first week after stepping up (say, from 5mg to 7.5mg). If you tolerate the increase well, you can continue with thigh or return to abdomen based on your preference.
This approach has worked well for many of our clients navigating dose escalation.
- How long should I try a new injection site before deciding if it’s better?
Give it at least two to three weeks - ideally a full month - before drawing conclusions.
Why patience matters:
- Week-to-week variation is normal - Side effects can fluctuate regardless of injection site
- Your body needs time to establish a pattern - One good or bad week doesn’t tell you much
- Other factors influence how you feel - Diet, stress, and activity levels all play a role
Keep a simple log noting your injection site, any side effects, and how you felt that week. After 3-4 weeks, review the pattern. If thigh injections consistently feel better than abdomen (or vice versa), you have useful information to guide your choice.
Systematic experimentation beats random switching.
- Does the injection site guidance differ for Zepbound compared to Mounjaro?
No - Zepbound and Mounjaro contain the same medication (tirzepatide), so all injection guidance is identical.
The only difference between them:
- Mounjaro - Approved for type 2 diabetes (and used off-label for weight loss)
- Zepbound - Approved specifically for chronic weight management
Same molecule, same injection sites, same technique, same considerations about site rotation. If you’re using Zepbound in the US or have seen both names mentioned, everything in this article applies equally to you.
References
U.S. Food and Drug Administration (2025a). Mounjaro (tirzepatide) injection, for subcutaneous use: Highlights of prescribing information.. U.S. Food and Drug Administration. accessdata.fda.gov/…25/215866s031lbl.pdf (Accessed: 2025-12-30)
U.S. Food and Drug Administration (2025b). Zepbound (tirzepatide) injection, for subcutaneous use: Highlights of prescribing information.. U.S. Food and Drug Administration. accessdata.fda.gov/…7806Orig1s020lbl.pdf (Accessed: 2025-12-30)
MedlinePlus (2024). Subcutaneous (SQ) injections.. U.S. National Library of Medicine. medlineplus.gov/…tructions/000430.htm (Accessed: 2025-12-30)
Eli Lilly and Company (2022). A study of tirzepatide (LY3298176) administered by injection in the abdomen, thigh, or upper arm in healthy participants (Clinical trial registration NCT04050670). ClinicalTrials.gov. clinicaltrials.gov/study/NCT04050670 (Accessed: 2025-12-30)
The Royal Children’s Hospital, Melbourne (2024). Nursing guidelines : Subcutaneous injections and device management. The Royal Children’s Hospital, Melbourne. rch.org.au/…d_device_management/
Eli Lilly and Company (2024). Mounjaro KwikPen (tirzepatide): Summary of Product Characteristics. Electronic Medicines Compendium (emc). medicines.org.uk/emc/product/15481/smpc (Accessed: 2025-12-30)
Medicines and Healthcare products Regulatory Agency (2024). Mounjaro (tirzepatide) - Product Information. MHRA Products. products.mhra.gov.uk/product/?product=MOUNJARO (Accessed: 2025-12-30)
Appendix
| Term | Meaning |
|---|---|
| AUC(0–∞) | Total exposure over time (area under the concentration–time curve from dosing to infinity). |
| Cmax | Peak blood level (highest measured concentration after dosing). |
| Geometric mean | An average suited to skewed concentration data; often closer to the ‘typical’ participant than an arithmetic mean. |
| Geometric CV (%) | How variable values were between participants (higher % = more variability). |
| Ratio (GLSM) | Model-based ratio of geometric least squares means (site ÷ abdomen, with abdomen as the reference). |
| CI | Confidence interval: the uncertainty range around the ratio estimate. |
| 0.80–1.25 bounds | Protocol comparability window: sites are considered similar if the 90% CI for the ratio lies entirely within 0.80–1.25. This is a common PK comparability convention; it does not mean the values are identical. |
| Injection site | N | Geometric mean AUC(0–∞) | Geometric CV (%) | Units |
|---|---|---|---|---|
| Abdomen | 54 | 112,000 | 24 | h·ng/mL |
| Upper arm | 54 | 111,000 | 21 | h·ng/mL |
| Thigh | 53 | 106,000 | 22 | h·ng/mL |
| Injection site | N | Geometric mean Cmax | Geometric CV (%) | Units |
|---|---|---|---|---|
| Abdomen | 54 | 603 | 25 | ng/mL |
| Upper arm | 54 | 556 | 23 | ng/mL |
| Thigh | 53 | 520 | 21 | ng/mL |
| Measure | Comparison | Ratio (GLSM) | CI level | CI (lower–upper) |
|---|---|---|---|---|
| AUC(0–∞) | Thigh vs abdomen | 0.953 | 90% | 0.935–0.970 |
| AUC(0–∞) | Upper arm vs abdomen | 0.990 | 90% | 0.972–1.010 |
| Cmax | Thigh vs abdomen | 0.862 | 90% | 0.818–0.909 |
| Cmax | Upper arm vs abdomen | 0.921 | 95%† | 0.874–0.971 |
| Abdomen is the reference site in all ratios. † ClinicalTrials.gov lists this CI as 95% for this comparison. | ||||
| Arm/Group Title | Tirzepatide - Abdomen | Tirzepatide - Upper Arm | Tirzepatide - Thigh | |||
|---|---|---|---|---|---|---|
| Affected / at Risk (%) | # Events | Affected / at Risk (%) | # Events | Affected / at Risk (%) | # Events | |
| Gastrointestinal disorders | ||||||
| Abdominal distension†1 | 8/54 (14.81%) | 8 | 10/54 (18.52%) | 10 | 8/53 (15.09%) | 8 |
| Constipation†1 | 4/54 (7.41%) | 4 | 2/54 (3.70%) | 3 | 2/53 (3.77%) | 2 |
| Diarrhoea†1 | 3/54 (5.56%) | 3 | 2/54 (3.70%) | 2 | 1/53 (1.89%) | 1 |
| Dyspepsia†1 | 10/54 (18.52%) | 11 | 11/54 (20.37%) | 13 | 6/53 (11.32%) | 7 |
| Eructation†1 | 3/54 (5.56%) | 3 | 4/54 (7.41%) | 5 | 1/53 (1.89%) | 1 |
| Flatulence†1 | 3/54 (5.56%) | 3 | 1/54 (1.85%) | 1 | 1/53 (1.89%) | 1 |
| Nausea†1 | 20/54 (37.04%) | 21 | 12/54 (22.22%) | 12 | 11/53 (20.75%) | 12 |
| Vomiting†1 | 9/54 (16.67%) | 10 | 10/54 (18.52%) | 10 | 4/53 (7.55%) | 4 |
| General disorders | ||||||
| Early satiety†1 | 2/54 (3.70%) | 2 | 0/54 (0.00%) | 0 | 3/53 (5.66%) | 3 |
| Metabolism and nutrition disorders | ||||||
| Decreased appetite†1 | 13/54 (24.07%) | 13 | 7/54 (12.96%) | 7 | 3/53 (5.66%) | 3 |
| Nervous system disorders | ||||||
| Headache†1 | 12/54 (22.22%) | 14 | 8/54 (14.81%) | 8 | 6/53 (11.32%) | 7 |
| † Indicates events were collected by systematic assessment 1 Term from vocabulary, MedDRA 22.0 | ||||||

